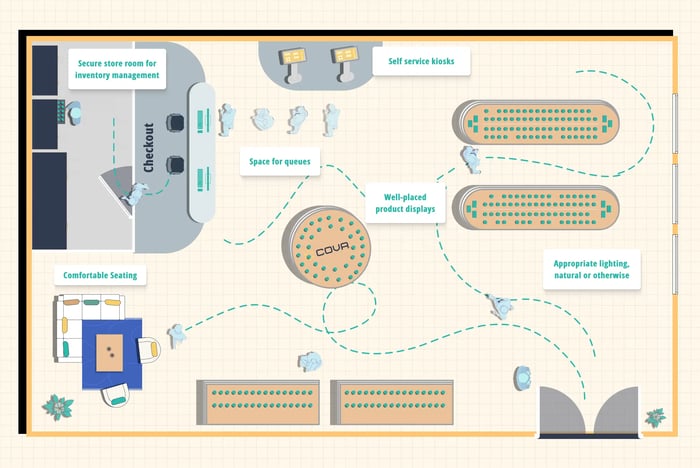
Best Practices
Marijuana Retail Report

Best Practices
Cannabis Payments Are Facing A Reckoning As Workarounds Disappear
Best Practices
Marijuana Retail Report
Best Practices
Mastering Cannabis Dispensary SEO: 28 Actionable Tips to Boost Online Visibility
-

 Cannabis News10 months ago
Cannabis News10 months ago“Our system can manage equipment across 10,000+ m² using just a few wires”
-

 Florida10 months ago
Florida10 months agoFlorida Workshop to Discuss What Constitutes a ‘Cartoon’ in Hemp Packaging
-

 Video8 months ago
Video8 months agoMazar-i-Sharif Hash Wednesday
-

 Video8 months ago
Video8 months agoAfghan Black Hash Wednesday
-

 aawh10 months ago
aawh10 months agoWeak Michigan Cannabis Sales Again in July – New Cannabis Ventures
-

 Video10 months ago
Video10 months agoFrom Finance to Wellness: Brad Zerman’s Impactful Pivot
-

 Video10 months ago
Video10 months agoDEA’s Cole Reverses His “priorities” ; Prohibitionists Dig In; Dead & Co Celebrates 60 years in SF
-

 Video10 months ago
Video10 months agoRe-release of the full show of Cannabis Coast to Coast news. Republican Texas DA Fires Up vs. laws;