“The issue is not whether the funds should be used, but how they are used and how we are doing it in a responsible and sustainable way.”
By Henry Culvyhouse, Mountain State Spotlight
This story was originally published by Mountain State Spotlight. Get stories like this delivered to your email inbox once a week; sign up for the free newsletter at https://mountainstatespotlight.org/newsletter.
Even with the veto he could have delayed it further $38 million spent on medical marijuana raised over the past four years, state Treasurer Larry Pack (R) now says he will release the funds during his original term.
Last week, Gov. Patrick Morrisey (R) vetoed a bill that would have required the release of medical marijuana funds to help the homeless and expedite child abuse and neglect cases in the court system. He said the bill tied up money for future expenses.
In his veto letter, Morrisey wrote, “West Virginia needs to do a better job of planning for the future, and cannot fully pre-commit future revenue like this if it has reserves to invest more in roads, water, sewer, site selection, rail and future tax cuts.”
Morrisey said he was willing to negotiate with the Legislature on how to spend the money.
“The issue is not whether the funds should be used, but how they are used and whether we are doing so responsibly and sustainably,” Lars Dalseide, a spokesman for the governor’s office, wrote in an email.
But the money was pre-committed in state code.
Pack’s office said 100 percent of that money will go to various offices and programs mandated by the original law; more than half to the Office of Medical Cannabis, with the remaining funds split between the substance abuse treatment grant program and law enforcement grants. The move negates the governor’s desire to use future reserves to deal with infrastructure and tax cuts.
In October, a Mountain State Spotlight investigation revealed that $34 million was deposited into an account held by the Treasury Department from the state’s medical marijuana program..
Pack’s office said the money it was not spent due to legal concerns about the drug. Currently, marijuana is listed as a Schedule I narcotic under federal law, meaning it has no medical use and is illegal.
Pack is not the first state treasurer to express concern. State Treasurer John Perdue (D) said his office would not keep money in 2018 after the Medical Cannabis Act was passed. Riley Moore (R), who beat Perdue in the 2020 race, never released the money.
In the 2026 Legislative Session, Del. Rep. Evan Worrell, R-Cabell, said he read a report on the funds raised and wanted to change it. He successfully led a bill that would have forced the state to spend money on a commission to help thousands of children with abuse and neglect in court and homelessness services.
Had the governor not vetoed the bill, the money would have been earmarked for one year for those things. The commission on substance abuse research, treatment, and abuse and neglect would continue for years to come.
Treasurer’s Office spokeswoman Carrie Smith said that due to the complexity of state and federal laws, the office had been working for months to release the money. He said that the money has been sent to the Department of Security and the Department of Health.
This the article appeared for the first time The focus of the Mountain State and is republished here under a Creative Commons Attribution-NoDerivs 4.0 International License.


 Cannabis News8 months ago
Cannabis News8 months ago
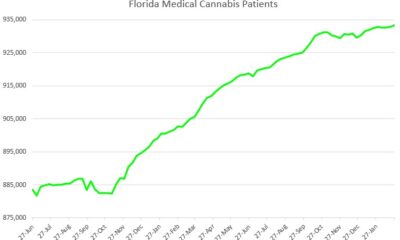
 Florida8 months ago
Florida8 months ago
 Video6 months ago
Video6 months ago
 Video8 months ago
Video8 months ago
 Video8 months ago
Video8 months ago
 aawh8 months ago
aawh8 months ago
 Video7 months ago
Video7 months ago
 Cannabis News8 months ago
Cannabis News8 months ago